The European Commission has adopted its Regulation to add 11 SVHCs to Annex XIV – the REACH authorisation list.
eur-lex.europa link: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX%3A32020R0171&from=GA&fbclid=IwAR1saipCr3iJX-V4h3sds6CP23U5H9TsGoXsbzj4eoW5fe_v648HXGOTLE0
The regulation for their inclusion was published in February 6, 2020 – COMMISSION REGULATION (EU) 2020/171. Their addition to the authorisation list increases the number of substances in the Annex XIV from 43 to 54.
Background
The authorisation process aims to ensure that substances of very high concern (SVHCs) are progressively replaced by less dangerous substances or technologies where technically and economically feasible alternatives are available.
The route to authorisation starts when a Member State or ECHA, at the request of the Commission, proposes a substance to be identified as an SVHC.More on obligations in terms of SVHCs on Ekotox: https://ekotox.eu/substances-of-very-high-concern-svhc/
The authorisation process is described in the scheme below:
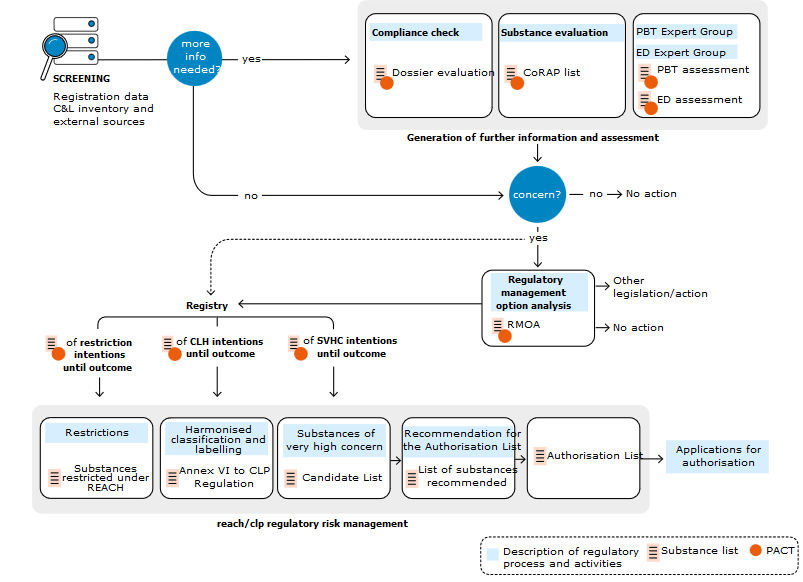
Link to ECHA: https://echa.europa.eu/substances-of-potential-concern
Substances included in the Authorisation List on 6 February 2020 and their SVHC properties:
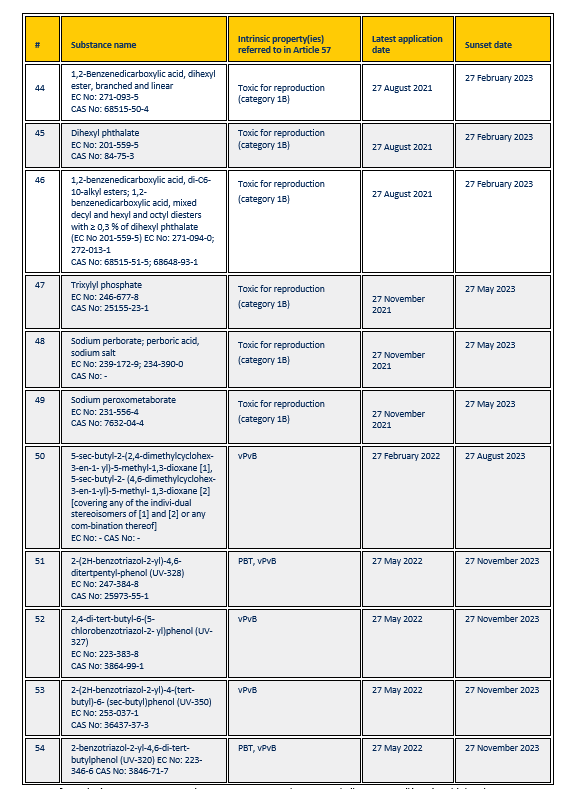
More information – REACH authorisation process on Ekotox: https://ekotox.eu/reach-authorisation/
![]()
Actual Ekotox webinars: www.ekotoxtraining.com
EKOTOX CENTERS
- Legislation and Regulatory Compliance Services (EU REACH, CLP, detergents, RoHS, WEEE, POPs…)
- Safety Data Sheets and products/mixtures registration on the EU member states markets (Poison Centers), UFI and more
- Biocides
- Cosmetics Safety
- Chemicals risk assessment / management
- Environmental Health & Safety & Risk Assessment
- Occupational Health & Safety & Risk Assessment
Ekotoxikologické centrum CZ
Centrum Ekotoksykologiczne (PL)
Ekotoxikologické centrum Bratislava, s.r.o. (SK)
Tomášikova 10/F, 821 03 Bratislava
Slovenská republika
Tel.: +421 (2) 4594 3712, 4594 5223
E-mail: ekotox(at)ekotox.eu
Web: https://ekotox.eu
FB: Ekotox Centers
Twitter: Ekotox Centers
Linkedin: Ekotox
