CLP SDS Services
EKOTOX CENTERS are providing consultancy services and customers support in the area of EU chemicals legislation
– REACH Registration, Only Representative, REACH Authorisation, REACH/CLP Screening, Safety Data Sheets, Legal Compliance Services, PCN and UFI services and more.
Contact us:
- E-mail: ekotox(at)ekotox.eu
- Tel +421 2 45943712
Safety data sheets are intended to provide the users of chemicals with the necessary information to help them protect human health and the environment.
Users of chemicals are companies or individuals within the European Union/European Economic Area. They use a substance, either on its own or in a mixture, in their industrial or professional activities.
Safety data sheets are intended both for the workers who handle the chemicals and for those responsible for safety.
A safety data sheet should be provided when:
1. A substance or a mixture is classified as hazardous;
2. A substance is persistent, bioaccumulative and toxic (PBT) or very persistent and very bioaccumulative (vPvB); or
3. A substance is included in the Candidate List for Authorisation according to REACH for other reasons than the above.
NEW REQUIREMENTS – CLP Regulation EU 2024/2865 Regulation – EU – 2024/2865 – EN – EUR-Lex
Regulation (EU) 2024/2865 of the European Parliament and of the Council of 23 October 2024 amending Regulation (EC) No 1272/2008 on classification, labelling and packaging of substances and mixtures.
The revised CLP Regulation ensures clear chemical labelling, particularly for online sales, and introduces simpler and clearer requirements so that chemicals can move freely across the EU.
The revision enhances chemical safety and information transparency:-
- Online stores will have to display hazardous properties clearly on their websites. This will protect consumers and ensure a level playing field for the industry.
- Labelling will be made simpler by allowing more flexible use of fold-out labels, introducing digital labelling and improving the legibility of labels.
- Advertisements and online offers will have to contain information on chemical hazards, facilitating informed consumer choices and the development of a market for sustainable consumer chemical products.
- For the first time, the safe sale of household chemicals via refill stations will be clarified. This will contribute to reducing packaging and packaging waste.
- There will be a more user-friendly inventory of substances notified by industry, benefiting SMEs.
- Explicit rules for classifying complex substances (those containing more than one constituent) will be introduced, while taking account of the specificities of natural complex substances, such as essential oils.
- Poison centres will receive more comprehensive information for medical emergencies, especially from cross-border distribution.
CLP Regulation
Regulation (EC) No 1272/2008 on classification, labelling and packaging of substances and mixtures (CLP) harmonises the provisions and criteria for the classification and labelling of
substances and mixtures within the Union. CLP taking into account the classification criteria and labelling rules of the UN Globally Harmonised System of Classification and Labelling of
Chemicals (GHS).
The CLP Regulation contributes to the UN GHS aim of describing and communicating the same hazards in the same way around the world.
The CLP Regulation entered into force on the 20 th of January 2009.
- On 12 April 2017, Commission Regulation (EU) 2017/54210 entered into force, adding a new Annex VIII to the CLP Regulation. It harmonises the information relating to emergency health
response that companies placing certain hazardous mixtures on the EU market are required to submit to the national appointed bodies. The information submitted needs to be consistent
with the information in the SDS. - the Unique Formula Identifier (UFI) required by the same Annex may need to be indicated in the SDS of certain hazardous mixtures.
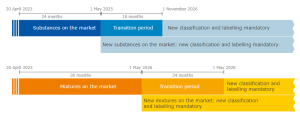
New hazard classes 2023
COMMISSION DELEGATED REGULATION (EU) 2023/707 of 19 December 2022 amending Regulation (EC) No 1272/2008 as regards hazard classes and criteria for the classification, labelling and packaging of substances and mixtures
Who should compile an SDS
The text of Annex II specifies in point 0.2.3 that:
“[…] The safety data sheet shall be prepared by a competent person who shall take into account the specific needs and knowledge of the user audience, as far as they are known.
Suppliers of substances and mixtures shall ensure that such competent persons have received appropriate training, including refresher training.”
Language(s) in which the SDS must be provided
According to REACH Article 31(5), “The safety data sheet shall be supplied in an official language of the Member State(s) where the substance or mixture is placed on the market,
unless the Member State(s) concerned provide otherwise”.
It should be noted that it is for the recipient Member State (MS) to provide otherwise – i.e. for example the existence of an exemption in the MS of manufacture does not give an exemption in a different MS where the substance or mixture is placed on the market.
Even if the MS provides otherwise, it may be desirable to always provide (potentially in addition) the SDS in the language of the country.
It should be noted that certain Member States require that the SDS be provided in additional official MS languages (of that MS, where there is more than one official language).
EKOTOX CENTERS – consultancy services and customers support in the area of EU chemicals legislation
– Safety Data Sheets authoring, revisions, complex management, Legal Compliance Services, PCN and UFI services and more.EKOTOX CENTERS – Contact us: https://ekotox.eu/about-company/contact-form/